Abstract
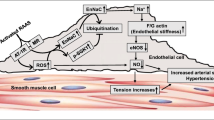
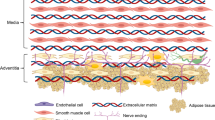
Many cardiovascular diseases present renin–angiotensin–aldosterone system (RAAS) hyperactivity as an important pathophysiological mechanism to be target in the therapeutic approaches. Moreover, arterial stiffness is currently considered as a new independent risk factor for cardiovascular disease in different clinical conditions, including hypertension and chronic kidney disease. In fact, excessive stimulation of angiotensin type 1 (AT1) receptors, as well as mineralocorticoid receptors, results in cellular growth, oxidative stress and vascular inflammation, which may lead to arterial stiffness and accelerate the process of vascular aging. In the last decades, a vasoprotective axis of the RAAS has been discovered, and now it is well established that new components with antioxidant and anti-inflammatory properties play important roles promoting vasodilation, natriuresis and reducing collagen deposition, thus attenuating arterial stiffness and improving endothelial function. In this review, we will focus on these pathophysiological mechanisms and the relevance of RAAS inhibition by different strategies to increase arterial compliance and to decelerate vascular aging.


Similar content being viewed by others
References
Zhuo JL, Ferrao FM, Zheng Y, Li XC. New frontiers in the intrarenal Renin-Angiotensin system: a critical review of classical and new paradigms. Front Endocrinol (Lausanne). 2013;4:1–14.
Te Riet L, Van Esch JHM, Roks AJM, Van Den Meiracker AH, Danser AHJ. Hypertension: renin-angiotensin-aldosterone system alterations. Circ Res. 2015;116:960–75.
Oparil S, Schmieder RE. New approaches in the treatment of hypertension. Circ Res. 2015;116:1074–95.
Safar ME, Levy BI, Struijker-Boudier H. Current perspectives on arterial stiffness and pulse pressure in hypertension and cardiovascular diseases. Circulation. 2003;107:2864–9.
Laurent S, Boutouyrie P, Asmar R, Gautier I, Laloux B, Guize L, et al. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension. 2001;37:1236–41.
Laurent S, Katsahian S, Fassot C, Tropeano AI, Gautier I, Laloux B, et al. Aortic stiffness is an independent predictor of fatal stroke in essential hypertension. Stroke. 2003;34:1203–6.
Vlachopoulos C, Aznaouridis K, Stefanadis C. Prediction of cardiovascular events and all-cause mortality with arterial stiffness: a systematic review and meta-analysis. J Am Coll Cardiol. 2010;55:1318–27.
Davies JI, Struthers AD. Pulse wave analysis and pulse wave velocity: a critical review of their strengths and weaknesses. J Hypertens. 2003;21:463–72.
Ong KT, Delerme S, Pannier B, Safar ME, Benetos A, Laurent S, Boutouyrie PI. Aortic stiffness is reduced beyond blood pressure lowering by short-term and long-term antihypertensive treatment: a meta-analysis of individual data in 294 patients. J Hypertens. 2011;29:1034–42.
Strazzullo P. Reducing sodium and increasing potassium intake. BMJ. 2013;346:f2195.
Strazzullo P, D’Elia L, Kandala N-B, Cappuccio FP. Salt intake, stroke, and cardiovascular disease: meta-analysis of prospective studies. BMJ. 2009;339:b4567.
Aburto NJ, Ziolkovska A, Hooper L, Elliott P, Cappuccio FP, Meerpohl JJ. Effect of lower sodium intake on health: systematic review and meta-analyses. BMJ. 2013;346:f1326.
Cook NR, Cutler JA, Obarzanek E, Buring JE, Rexrode KM, Kumanyika SK, et al. Long term effects of dietary sodium reduction on cardiovascular disease outcomes: observational follow-up of the trials of hypertension prevention (TOHP). BMJ. 2007;334:885–8.
Edwards DG, Farquhar WB. Vascular effects of dietary salt. Curr Opin Nephrol Hypertens. 2015;24:8–13.
D’Elia L, Galletti F, La Fata E, Sabino P, Strazzullo P. Effect of dietary sodium restriction on arterial stiffness: systematic review and meta-analysis of the randomized controlled trials. J Hypertens. 2017;35:1–10.
Todd AS, Macginley RJ, Schollum JB, Johnson RJ, Williams SM, Sutherland WH, et al. Dietary salt loading impairs arterial vascular reactivity. Am J Clin Nutr. 2010;91:557–64.
Frohlich ED. The salt conundrum: a hypothesis. Hypertension. 2007;50:161–6.
Matavelli LC, Zhou X, Varagic J, Susic DFE. Salt loading produces severe renal hemodynamic dysfunction independent of arterial pressure in spontaneously hypertensive rats. Am J Physiol Hear Circ Physiol. 2007;292:H814–9.
Leenen FH, Ruzicka M, Huang BS. The brain and salt-sensitive hypertension. Curr Hypertens Rep. 2002;4:129–35.
Schmidlin O, Forman A, Sebastian AMRJ. Sodium-selective salt sensitivity: its occurrence in blacks. Hypertension. 2007;50:1085–92.
Osborn JWFG. Region specific changes in sympathetic nerve activity in AngII-salt hypertension. Exp Physiol. 2010;95:61–8.
Stocker SD, Madden CJSA. Excess dietary salt intake alters the excitability of central sympathetic networks. Physiol Behav. 2010;100:519–24.
Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352:1685–95.
Mäki-Petäjä KM, Wilkinson IB. Inflammation and large arteries: potential mechanisms for inflammation-induced arterial stiffness. Artery Res. 2012;6:59–64.
Ayhan H, Kasapkara HA, Aslan AN, Durmaz T, Keleş T, Akçay M, et al. Relationship of neutrophil-to-lymphocyte ratio with aortic stiffness in type 1 diabetes mellitus. Can J Diabetes. 2015;39:317–21.
Avolio A, Butlin M, Liu YY, Viegas K, Avadhanam B, Lindesay G. Regulation of arterial stiffness: cellular, molecular and neurogenic mechanisms. Artery Res. 2011;5:122–7.
McEniery CM, Qasem A, Schmitt M, Avolio AP, Cockcroft JR, Wilkinson IB. Endothelin-1 regulates arterial pulse wave velocity in vivo. J Am Coll Cardiol. 2003;42:1975–81.
Lacolley P, Challande P, Regnault V, Lakattac EG, Wang M. Cellular and molecular determinants of arterial aging. In: Nilsson P, Olsen M, Laurent S, editors. Early Vascular Aging. Elsevier, Academic Press; 2015. p. 7–13.
Wang M, Jiang L, Monticone RE, Lakatta EG. Proinflammation: the key to arterial aging. Trends Endocrinol Metab. 2014;25:72–9.
Kranzhofer R, Schmidt J, Pfeiffer CA, Hagl S, Libby P, Kubler W. Angiotensin induces inflammatory activation of human vascular smooth muscle cells. Arterioscler Thromb Vasc Biol. 1999;19:1623–9.
Hahn AW, Jonas U, Buhler FR, Resink TJ. Activation of human peripheral monocytes by angiotensin II. FEBS Lett. 1994;347:178–80.
Ferrario CM, Strawn WB. Role of the renin-angiotensin aldosterone system and proinflammatory mediators in cardiovascular disease. Am J Cardiol. 2006;98:121–8.
Martinez FA. Aldosterone inhibition and cardiovascular protection: more important than it once appeared. Cardiovasc Drugs Ther. 2010;24:345–50.
Cascella T, Radhakrishnan Y, Maile LA, Walker H, Busby J, Gollahon K, et al. Aldosterone enhances IGF-I-mediated signaling and biological function in vascular smooth muscle cells. Endocrinology. 2010;151:5851–64.
Nam SH, Kang SG, Song SW. The neutrophil-lymphocyte ratio is associated with coronary artery calcification in asymptomatic Korean males: a crosssectional study. Biomed Res Int. 2017;2017:1989417.
Balta S, Kurtoglu E, Kucuk U, Demirkol S, Öztürk C. Neutrophil-lymphocyte ratio as an important assessment tool. Expert Rev Cardiovasc Ther. 2014;12:537–8.
Karaman M, Balta S, Seyit Ahmet AY, Cakar M, Naharci I, Demirkol S, et al. The comparative effects of valsartan and amlodipine on vWf levels and N/L ratio in patients with newly diagnosed hypertension. Clin Exp Hypertens. 2013;35:516–22.
Horne BD, Anderson JL, John JM, Weaver A, Bair TL, Jensen KR, et al. Which white blood cell subtypes predict increased cardiovascular risk? J Am Coll Cardiol. 2005;45:1638–43.
Pacurari M, Kafoury R, Tchounwou PB, Ndebele K. The renin–angiotensin–aldosterone system in vascular inflammation and remodeling. Int J Inflam. 2014;2014:689360.
Harvey A, Montezano AC, Touyz RM. Vascular biology of ageing—implications in hypertension. J Mol Cell Cardiol. 2015;83:112–21.
Guzik TJ, Touyz RM. Oxidative stress, inflammation, and vascular aging in hypertension. Hypertension. 2017;70:660–7.
Mikhed Y, Daiber A, Steven S. Mitochondrial oxidative stress, mitochondrial DNA damage and their role in age-related vascular dysfunction. Int J Mol Sci. 2015;16:15918–53.
Kokoszka JE, Coskun P, Esposito LA, Wallace DC. Increased mitochondrial oxidative stress in the Sod2 (±) mouse results in the age-related decline of mitochondrial function culminating in increased apoptosis. Proc Natl Acad Sci USA Natl Acad Sci. 2001;98:2278–83.
Konior A, Schramm A, Czesnikiewicz-Guzik M, Guzik TJ. NADPH oxidases in vascular pathology. Antioxid Redox Signal. 2014;20:2794–814.
Ungvari Z, Bailey-Downs L, Sosnowska D, Gautam T, Koncz P, Losonczy G, et al. Vascular oxidative stress in aging: a homeostatic failure due to dysregulation of NRF2-mediated antioxidant response. AJP Hear Circ Physiol. 2011;301:H363–72.
Förstermann U, Li H. Therapeutic effect of enhancing endothelial nitric oxide synthase (eNOS) expression and preventing eNOS uncoupling. Br J Pharmacol. 2011;164:213–23.
Mikolajczyk TP, Nosalski R, Szczepaniak P, Budzyn K, Osmenda G, Skiba D, et al. Role of chemokine RANTES in the regulation of perivascular inflammation, T-cell accumulation, and vascular dysfunction in hypertension. FASEB J. 2016;30:1987–99.
Leong X-F, Mustafa MR, Das S, Jaarin K. Association of elevated blood pressure and impaired vasorelaxation in experimental Sprague-Dawley rats fed with heated vegetable oil. Lipids Health Dis. 2010;9:66.
Yung L-M, Sánchez-Duffhues G, ten Dijke P, Yu PB. Bone morphogenetic protein 6 and oxidized low-density lipoprotein synergistically recruit osteogenic differentiation in endothelial cells. Cardiovasc Res. 2015;108:278–87.
Brown KA, Didion SP, Andresen JJ, Faraci FM. Effect of aging, MnSOD deficiency, and genetic background on endothelial function: evidence for MnSOD haploinsufficiency. Arterioscler Thromb Vasc Biol Am Heart Assoc. 2007;27:1941–6.
Ferrario CM. Addressing the theoretical and clinical advantages of combination therapy with inhibitors of the renin-angiotensin-aldosterone system: antihypertensive effects and benefits beyond BP control. Life Sci. 2010;86:289–99.
Yusuf S, Teo KK, Pogue J, Dyal L, Copland I, Schumacher H, et al. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358:1547–59.
Rhaleb NE, Yang XP, Carretero OA. The Kallikrein-Kinin system as a regulator of cardiovascular and renal function. Compr Physiol. 2011;1:971–93.
Linz W, Wiemer G, Gohlke P, Unger T, Scholkens B. Contribution of kinins to the cardiovascular actions of angiotensin-converting enzyme inhibitors. Pharmacol Rev. 1995;47:25–49.
Düsing R. Mega clinical trials which have shaped the RAS intervention clinical practice. Ther Adv Cardiovasc Dis. 2016;10:133–50.
Wong MCS, Chan DKL, Wang HHX, Tam WWS, Cheung CSK, Yan BP, et al. The incidence of all-cause, cardiovascular and respiratory disease admission among 20,252 users of lisinopril vs. perindopril: a cohort study. Int J Cardiol. 2016;219:410–6.
Shahin Y, Khan JA, Chetter I. Angiotensin converting enzyme inhibitors effect on arterial stiffness and wave reflections: a meta-analysis and meta-regression of randomised controlled trials. Atherosclerosis. 2012;221:18–33.
Van Vark LC, Bertrand M, Akkerhuis KM, Brugts JJ, Fox K, Mourad JJ, et al. Angiotensin-converting enzyme inhibitors reduce mortality in hypertension: a meta-analysis of randomized clinical trials of renin-angiotensin-aldosterone system inhibitors involving 158 998 patients. Eur Heart J. 2012;33:2088–97.
Intengan HD, Thibault G, Li JS, Schiffrin EL. Resistance artery mechanics, structure, and extracellular components in spontaneously hypertensive rats: effects of angiotensin receptor antagonism and converting enzyme inhibition. Circulation. 1999;100:2267–75.
Wojakowski W, Gminski J, Siemianowicz K, Goss M, Machalski M. The influence of angiotensin-converting enzyme inhibitors on the aorta elastin metabolism in diet-induced hypercholesterolaemia in rabbits. J Renin Angiotensin Aldosterone Syst. 2001;2:37–42.
Carey RM. Update on angiotensin AT2 receptors. Curr Opin Nephrol Hypertens. 2017;26:91–6.
Carey RM. AT2 receptors: potential therapeutic targets for hypertension. Am J Hypertens. 2017;30:339–47.
Lévy BI. Can angiotensin II Type 2 receptors have deleterious effects in cardiovascular disease? Implications for therapeutic blockade of the renin-angiotensin system. Circulation. 2004;109:8–13.
Ichihara S, Senbonmatsu T, Price E, Ichiki T, Gaffney FA, Inagami T. Angiotensin II type 2 receptor is essential for left ventricular hypertrophy and cardiac fibrosis in chronic angiotensin II-induced hypertension. Circulation. 2001;104:346–51.
Senbonmatsu T, Ichihara S, Price E, Gaffney FA, Inagami T. Evidence for angiotensin II type 2 receptor–mediated cardiac myocyte enlargement during in vivo pressure overload. J Clin Invest. 2000;106:R25–9.
Briet M, Schiffrin EL. Vascular actions of aldosterone. J Vasc Res. 2013;50:89–99.
Briet M, Barhoumi T, Mian MOR, Coelho SC, Ouerd S, Rautureau Y, et al. Aldosterone-induced vascular remodeling and endothelial dysfunction require functional angiotensin type 1a receptors novelty and significance. Hypertension. 2016;67:897–905.
Druppel V, Kusche-Vihrog K, Grossmann C, Gekle M, Kasprzak B, Brand E, et al. Long-term application of the aldosterone antagonist spironolactone prevents stiff endothelial cell syndrome. FASEB J. 2013;27:3652–9.
Silva MAB, Cau SBA, Lopes RAM, Manzato CP, Neves KB, Bruder-Nascimento T, et al. Mineralocorticoid receptor blockade prevents vascular remodelling in a rodent model of type 2 diabetes mellitus. Clin Sci. 2015;129:533–45.
Savoia C, Touyz RM, Amiri F, Schiffrin EL. Selective mineralocorticoid receptor blocker eplerenone reduces resistance artery stiffness in hypertensive patients. Hypertension. 2008;51:432–9.
Mahmud A, Feely J. Aldosterone-to-renin ratio, arterial stiffness, and the response to aldosterone antagonism in essential hypertension. Am J Hypertens. 2005;18:50–5.
White WB, Duprez D, St Hillaire R, Krause S, Roniker B, Kuse-Hamilton J, et al. Effects of the selective aldosterone blocker eplerenone versus the calcium antagonist amlodipine in systolic hypertension. Hypertension. 2003;41:1021–6.
Hwang M-H, Yoo J-K, Luttrell M, Kim H-K, Meade TH, English M, et al. Role of mineralocorticoid receptors in arterial stiffness in human aging. Exp Gerontol. 2013;48:701–4.
Nobakht N, Kamgar M, Rastogi A, Schrier RW. Limitations of angiotensin inhibition. Nat Rev Nephrol. 2011;7:356–9.
O’Brien E, Barton J, Nussberger J, Mulcahy D, Jensen C, Dicker P, et al. Aliskiren reduces blood pressure and suppresses plasma renin activity in combination with a thiazide diuretic, an angiotensin-converting enzyme inhibitor, or an angiotensin receptor blocker. Hypertension. 2007;49:276–84.
Virdis A, Ghiadoni L, Qasem AA, Lorenzini G, Duranti E, Cartoni G, et al. Effect of aliskiren treatment on endothelium-dependent vasodilation and aortic stiffness in essential hypertensive patients. Eur Heart J. 2012;33:1530–8.
McMurray JJV, Krum H, Abraham WT, Dickstein K, Køber LV, Desai AS, et al. Aliskiren, Enalapril, or Aliskiren and enalapril in heart failure. N Engl J Med. 2016;374:1521–32.
Solinski HJ, Gudermann T, Breit A. Pharmacology and signaling of MAS-related G protein-coupled receptors. Pharmacol Rev. 2014;66:570–97.
Santos RAS, e Silva ACS, Maric C, Silva DMR, Machado RP, de Buhr L, et al. Angiotensin-(1-7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc Natl Acad Sci. 2003;100:8258–63.
Ferrario CM. Angiotensin-converting enzyme 2 and angiotensin-(1-7): an evolving story in cardiovascular regulation. Hypertension. 2006;47:515–21.
Chappell MC. Emerging evidence for a functional angiotensin-converting enzyme 2-angiotensin-(1-7)-MAS receptor axis: more than regulation of blood pressure? Hypertension. 2007;50:596–9.
Danilczyk U, Penninger JM. Angiotensin-converting enzyme II in the heart and the kidney. Circ Res. 2006;98:463–71.
Li XC, Zhang J, Zhuo JL. The vasoprotective axes of the renin-angiotensin system: physiological relevance and therapeutic implications in cardiovascular, hypertensive and kidney diseases. Pharmacol Res. 2017;125:21–38.
Passos-Silva DG, Brandan E, Santos RAS. Angiotensins as therapeutic targets beyond heart disease. Trends Pharmacol Sci. 2015;36:310–20.
Chappell MC. Biochemical evaluation of the renin-angiotensin system: the good, bad, and absolute? Am J Physiol Hear Circ Physiol. 2016;310:H137–52.
Kaschina E, Namsolleck P, Unger T. AT2 receptors in cardiovascular and renal diseases. Pharmacol Res. 2017;125:39–47.
Li XC, Campbell DJ, Ohishi M, Yuan S, Zhuo JL. AT1 receptor-activated signaling mediates angiotensin IV-induced renal cortical vasoconstriction in rats. Am J Physiol Ren Physiol. 2006;290:F1024–33.
Kemp BA, Howell NL, Gildea JJ, Keller SR, Padia SH, Carey RM. AT2 receptor activation induces natriuresis and lowers blood pressure. Circ Res. 2014;115:388–99.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Human participants and/or animals rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
This article is a Review and informed consent is not applicable.
Additional information
This article is part of the topical collection on Vascular Aging and Arterial Stiffness.
Rights and permissions
About this article
Cite this article
Neves, M.F., Cunha, A.R., Cunha, M.R. et al. The Role of Renin–Angiotensin–Aldosterone System and Its New Components in Arterial Stiffness and Vascular Aging. High Blood Press Cardiovasc Prev 25, 137–145 (2018). https://doi.org/10.1007/s40292-018-0252-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40292-018-0252-5